Bibliography Background About KRIS
Dissolved Oxyggen
 Oxygen is one of several dissolved gases important to aquatic systems. Dissolved oxygen is necessary to maintain aerobic conditions in surface waters and is considered a primary indicator when assessing the suitability of surface waters to support aquatic life. The oxygen content of natural waters varies with temperature, salinity, turbulence, photosynthetic activity of algae and plants, and atmospheric pressure. Primary sources of oxygen in water bodies include diffusion of atmospheric oxygen across the air-water interface and photosynthesis of aquatic plants.
Oxygen is one of several dissolved gases important to aquatic systems. Dissolved oxygen is necessary to maintain aerobic conditions in surface waters and is considered a primary indicator when assessing the suitability of surface waters to support aquatic life. The oxygen content of natural waters varies with temperature, salinity, turbulence, photosynthetic activity of algae and plants, and atmospheric pressure. Primary sources of oxygen in water bodies include diffusion of atmospheric oxygen across the air-water interface and photosynthesis of aquatic plants.
For maintenance of aquatic health, dissolved oxygen concentrations should approach saturation – that concentration which is in equilibrium with the partial pressure of atmospheric oxygen. Solubility of oxygen is a function of water temperature, salinity, and atmospheric pressure; decreasing with rising temperature and salinity, and increasing with rising atmospheric pressure. Freshwater at sea level has a saturation dissolved oxygen concentration of about 14.6 mg/l at 0C (32F) and 8.2 mg/l at 25C (77F).
Generally, oxygen concentrations are below saturation due to the presence and oxidation of decaying organic matter (suspended, benthic, or sediment). In addition to the organic, or carbonaceous oxygen demand, nitrogenous materials may exert an oxygen demand through bacterial oxidation of ammonia to nitrate (Krenkel and Novotny, 1980). Other materials may likewise produce an oxygen demand on the system. Thus, variations can occur seasonally as well as over 24-hour periods in response to temperature and biological activity. Concentrations below 5 mg/l may adversely affect function and survival of biological communities, and below 2 mg/l can lead to death of most fishes (Water Quality Assessments, 1996). Dissolved oxygen distribution in reservoirs may vary substantially from river systems due to differing hydraulic regimes.
Primary production (algae) acts as both a source and sink of dissolved oxygen. During daylight hours algal photosynthesis produces oxygen in excess of algal demands (respiration), often resulting in dissolved oxygen levels in excess of saturation, i.e., super-saturation. During nighttime periods when photosynthesis is absent, algal respiration may reduce dissolved oxygen levels significantly. Another mechanism that may increase or decrease dissolved oxygen is transfer through the air-water interface. Typically the transfer is from the atmosphere into the water (re-aeration) because dissolved oxygen in most natural waters is below saturation. However, under supersaturated conditions there is a net transfer of oxygen from the water body to the atmosphere.
Usually, dissolved oxygen levels in excess of 7.0 mg/l are desired to maintain aquatic ecosystem health.
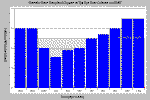 Dissolved oxygen may fluctuate dramatically when algae blooms are occurring. The chart at left shows dissolved oxygen dropping to 3.1 milligrams per liter (mg/l) at 3:00 A.M. but rising to suitable levels for salmonids during the day. The night time dissolved oxygen sag is due to respiration of algae while photosynthesis during the day produces oxygen. Chart from Klamath-Trinity KRIS Version 2.0. Data from U.S. Fish and Wildlife Service, Arcata.
Dissolved oxygen may fluctuate dramatically when algae blooms are occurring. The chart at left shows dissolved oxygen dropping to 3.1 milligrams per liter (mg/l) at 3:00 A.M. but rising to suitable levels for salmonids during the day. The night time dissolved oxygen sag is due to respiration of algae while photosynthesis during the day produces oxygen. Chart from Klamath-Trinity KRIS Version 2.0. Data from U.S. Fish and Wildlife Service, Arcata.
Impacts on Anadromous Salmonids: Dissolved Oxygen
Dissolved oxygen is one of the limiting factors for fish. In general, aquatic organisms possess highly specialized gas exchange systems that allow maximum utilization of available oxygen. Specifically, salmonids require a sufficient oxygen gradient, (oxygen tension gradient) between their bodies and surrounding waters to allow gas (i.e., dissolved oxygen) exchange through diffusion across the gills and into the blood. Further, there must be sufficient available oxygen to fulfill minimum metabolic demands – maintenance of minimum bodily functions (Davis 1975).

Theoretically, a critical oxygen level for each species exists (Colt et al, 1979); however limited data is available. Fish can resist or tolerate short-term oxygen reductions. It has been determined that certain species may acclimatize to reduced dissolved oxygen levels, as observed in trout species, if declines are not abrupt (Vinson and Levesque 1994, Davis 1975). Behaviorally, fish may avoid low dissolved oxygen conditions by physically moving out of an area. Finally, low oxygen levels can also increase toxicity of contaminants to anadromous fish, including ammonia, zinc, lead, and copper (Colt et al 1979, Davis 1975).
Excessive oxygen levels appear to have no deleterious impact on fish. Concentration levels of 250 to 300 percent of saturation are not lethal (Wiebe and McGavock 1932). This condition should not be confused with gas bubble disease, the result of local gas supersaturation (typically nitrogen) caused by air entrainment, heating of water, and air vented into power turbines (Colt et al 1979).
Effects of Dissolved Oxygen on Adult Salmonids: Migration and Spawning
 Recommended oxygen levels for all spawning salmonids is a minimum of 80 percent saturation with temporary levels no lower than 5mg/l. While maximum sustained swimming speeds of adult coho were adversely affected when dissolved oxygen was reduced below saturation at temperatures from 10°C (50°F) to 20°C (68°F) (Reiser and Bjornn 1979). Though different from steelhead, adult rainbow trout show negative effects at concentrations less than about 5 mg/l or 50% saturation, including elevated breathing amplitude, reduced heart rate, and reduced swimming speeds (Vinson and Levesque 1994).
Recommended oxygen levels for all spawning salmonids is a minimum of 80 percent saturation with temporary levels no lower than 5mg/l. While maximum sustained swimming speeds of adult coho were adversely affected when dissolved oxygen was reduced below saturation at temperatures from 10°C (50°F) to 20°C (68°F) (Reiser and Bjornn 1979). Though different from steelhead, adult rainbow trout show negative effects at concentrations less than about 5 mg/l or 50% saturation, including elevated breathing amplitude, reduced heart rate, and reduced swimming speeds (Vinson and Levesque 1994).
Chinook, coho, and steelhead have exhibited avoidance behavior of low dissolved oxygen water (Warren et al 1973). Juvenile chinook salmon strongly avoided low DO water (1.5 mg/l to 4.5 mg/l) areas during summer periods when temperatures were high, but did not exhibit similar behavior in fall when water temperatures were cooler (Whitmore 1960). Matthews and Berg (1996) explored the relationship between temperature and dissolved oxygen for rainbow trout: when faced between high temperature and low dissolved oxygen, trout typically were distributed closest to the water with the lowest temperature despite low oxygen content. Davis (1975) categorized response of freshwater salmonids into function without impairment, exhibition of initial oxygen distress, and significant portion of population affected. The results are presented in Table 3.6.
These criteria were developed to ensure adequate oxygen tension gradient to transfer oxygen to the blood as well as sufficient oxygen to fulfill metabolic requirements. At low temperatures where solubility of oxygen is high, the criteria were set to ensure oxygen tension gradient criteria were met. At high temperatures, higher saturation values were required to provide sufficient metabolic processes. Thus, the percent saturation values more effectively represent anadromous fish requirements than the mass per volume (mg/l) criteria. The reader is referred to Davis (1975) for additional details.
Effect of Dissolved Oxygen on Egg incubation and Juvenile Rearing
 Reiser and Bjornn (1979) report that low dissolved oxygen concentrations during egg incubation may delay hatching, increase anomalous development, stimulate premature hatching, and ultimately lead to weaker, smaller fry. They further state that coho salmon and steelhead survival drops off dramatically when intragravel dissolved oxygen concentration falls below an average of about 8 mg/l. Although dissolved oxygen requirements for successful incubation are species and developmental stage dependent, they suggest minimum concentrations at or near saturation with temporary reductions no lower than 5 mg/l for anadromous salmonids. Similar to adult dissolved oxygen criteria, Davis (1975) categorizes dissolved oxygen criteria for salmonid larvae and mature eggs based on oxygen tension gradient and metabolic requirements, both in mg/l and as percent saturation. The results are presented in Table 3.7. As with Table 3.6, the percent saturation values account for required oxygen tension gradient and sufficient oxygen.
Reiser and Bjornn (1979) report that low dissolved oxygen concentrations during egg incubation may delay hatching, increase anomalous development, stimulate premature hatching, and ultimately lead to weaker, smaller fry. They further state that coho salmon and steelhead survival drops off dramatically when intragravel dissolved oxygen concentration falls below an average of about 8 mg/l. Although dissolved oxygen requirements for successful incubation are species and developmental stage dependent, they suggest minimum concentrations at or near saturation with temporary reductions no lower than 5 mg/l for anadromous salmonids. Similar to adult dissolved oxygen criteria, Davis (1975) categorizes dissolved oxygen criteria for salmonid larvae and mature eggs based on oxygen tension gradient and metabolic requirements, both in mg/l and as percent saturation. The results are presented in Table 3.7. As with Table 3.6, the percent saturation values account for required oxygen tension gradient and sufficient oxygen.
Maximum growth for juvenile coho occurs at about 8.3 mg/l (Colt et al 1979). Herrmann et al (1962) reported that the growth of juvenile coho salmon maintained at 20°C declined slightly when concentrations were reduced from 8 mg/l to 5 mg/l, while growth declined more rapidly at lower concentrations. Mortality was high at levels averaging 2.1 to 2.3 mg/l, and those surviving showed a reduced consumption and weight loss (Colt et al 1979).
References
Colt, J., S. Mitchell, G. Tchobanoglous, and A. Knight. 1979. The use and potential for aquatic species for wastewater treatment: Appendix B, the environmental requirements of fish. Publication No. 65, California State Water Resources Control Board, Sacramento, CA.
Davis, J.C. 1975. Minimal dissolved oxygen requirements of aquatic life with emphasis on Canadian species: a review. Journal of Fisheries Research Board Canada. 32(12), 2295-2332.
Deas, M.L. and G.T. Orlob. 1999. Klamath River Modeling Project. Project #96-HP-01. Assessment of Alternatives for Flow and Water Quality Control in the Klamath River below Iron Gate Dam. University of California Davis Center for Environmental and Water Resources Engineering. Report No. 99-04. Report 236 pp.
Krenkel, P.A. and V. Novotney. 1980. Water Quality Management. Academic Press, New York.
North Coast Regional Water Quality Control Board. 2001. Water Quality Control Plan for the North Coast Region. Staff report adopted by the North Coast Regional Water Quality Control Board on June 28, 2001. Santa Rosa, CA. 124 p. Appendix.
United States Environmental Protection Agency (EPA). 1973. Development of Dissolved Oxygen Criteria for Freshwater Fish. Ecological Research Series EPA-R3-73-019. February.
Water Quality Assessments. 1996. Water Quality assessments: A guide to the use of biota, sediments and water in environmental modeling. Ed. D. Chapman. Published on behalf of UNESCO United Nations Education, Scientific, and Cultural Organization; WHO World Health Organization; UNEP United Nations Environmental Program. Chapman & Hall, London.